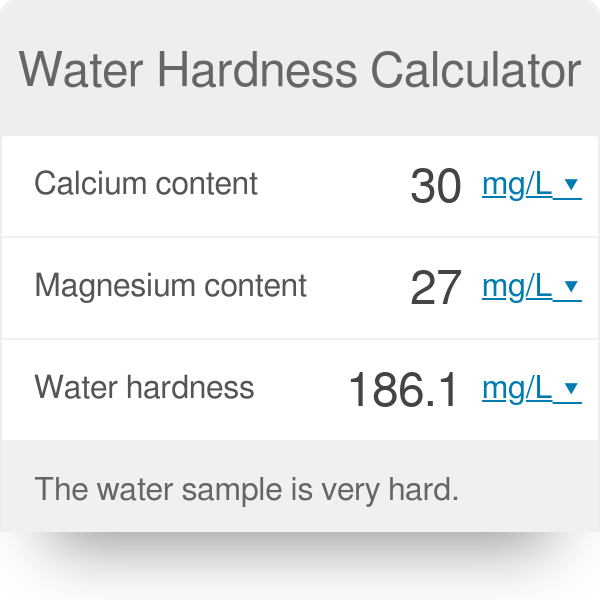
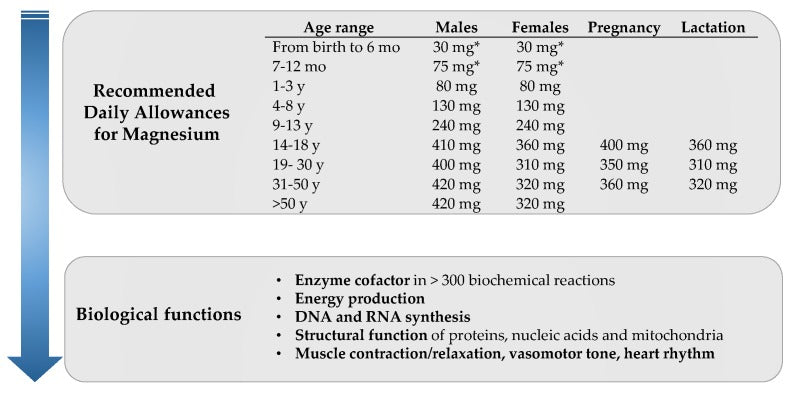
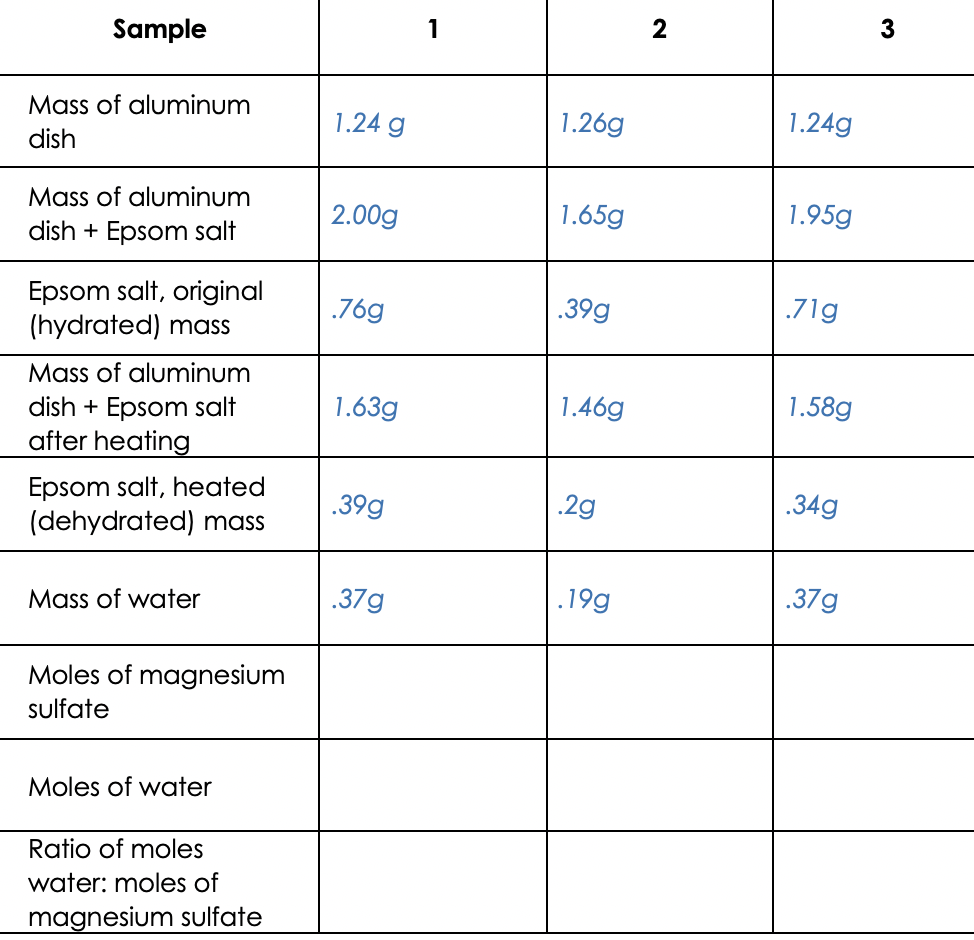
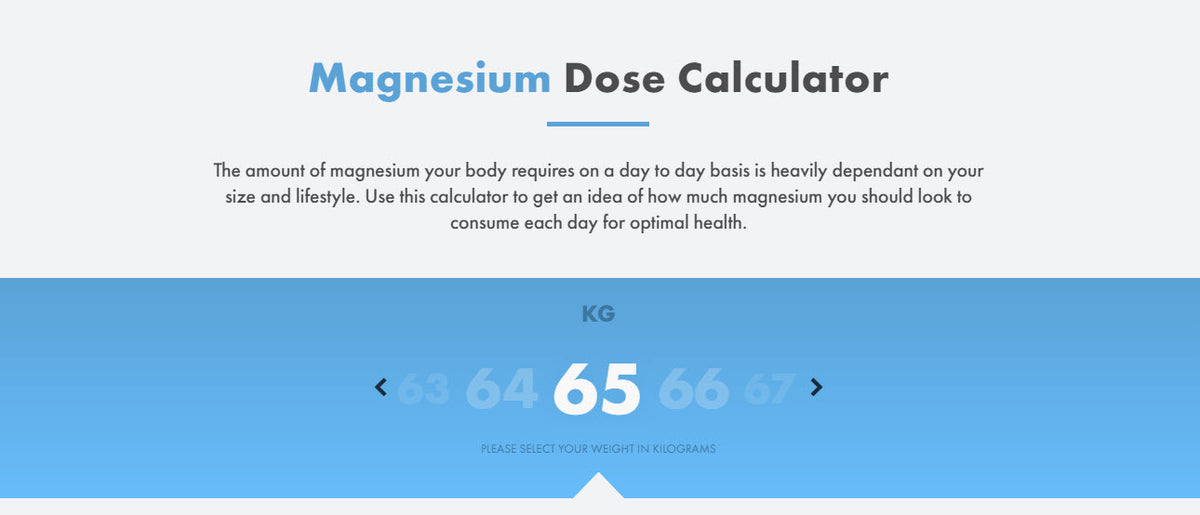
View question - Calculate the mass of magnesium oxide formed when 0.52g of magnesium is burned according to the following equation: 2 Mg^s + O^2 ---> 2 MgO^s
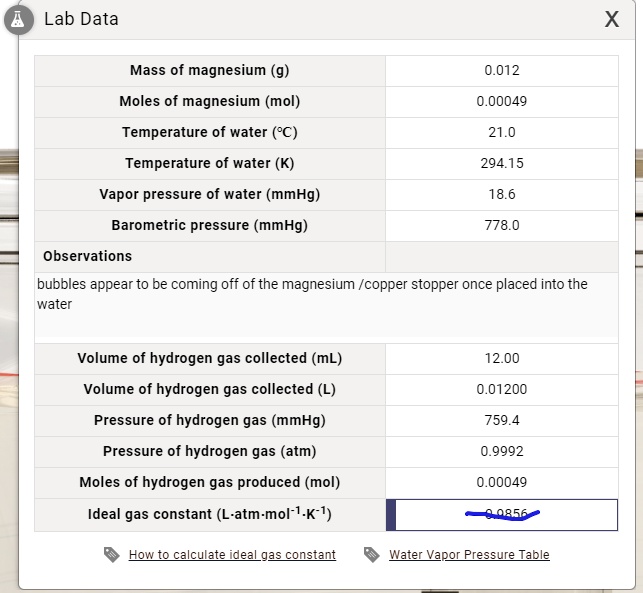
SOLVED: Lab Data X Mass of magnesium (g) 0.012 Moles of magnesium (mol) 0.00049 Temperature of water (PC) 21.0 Temperature of water (K) 294.15 Vapor pressure of water (mmHg) Barometric pressure (mmHg)

Average atomic mass of magnesium is `24.31`amu. This magnesium is composed of 79 mole % of `24mg... - YouTube

SOLVED: 'Magnesium reacts with iron chloride solution. Calculate the mass of iron produced in mg Question Points) Magnesium reacts with iron chloride solution: 3 Mg 2 FeCla 2 Fe 3 MgClz 0.120

SOLVED: Calculate the number of magnesium atoms in a 60.0 g sample of forsterite (MgSio4) Be sure your answer has unit symbol if necessary, and round it to 3 significant digits. X1O
![Calculate the number of molecules present in 0.5 moles of magnesium oxide (MgO) . [Atomic weights : Mg = 24, O = 16 ] Calculate the number of molecules present in 0.5 moles of magnesium oxide (MgO) . [Atomic weights : Mg = 24, O = 16 ]](https://dwes9vv9u0550.cloudfront.net/images/2527137/b4ebf9c0-6b64-43b4-94e9-555f3180cc58.jpg)